Mental health innovation is expanding quickly. That includes digital therapy apps, device-based treatments like TMS, and newer medications like esketamine — many of which have received FDA approval or clearance in recent years but are still relatively new to patients.
But new survey data shows many people remain cautious about trying unfamiliar mental health treatments, often weighing safety, clinical evidence, and cost before deciding whether to explore them. In a national survey of 487 U.S. adults with depression and/or anxiety, conducted by YouGov for Grow Therapy in February 2026, patients expressed openness to certain innovations, while remaining more cautious about complex or unfamiliar treatments.
Relatively few survey respondents had actually tried newer treatment formats, which reflects something broader than a lack of interest. Low adoption doesn’t necessarily indicate little interest in or value of new therapeutic technology. Instead, it reflects a broader reality: FDA approval is only the starting point. Once a treatment is approved or cleared, patients and providers still need to evaluate whether it’s clinically appropriate for that person’s needs and realistically accessible given cost and coverage.
Deciding which tools or treatments belong in a care plan requires more than awareness. It requires clinical guidance, evidence review, and a clear-eyed look at what’s actually accessible.
Key takeaways
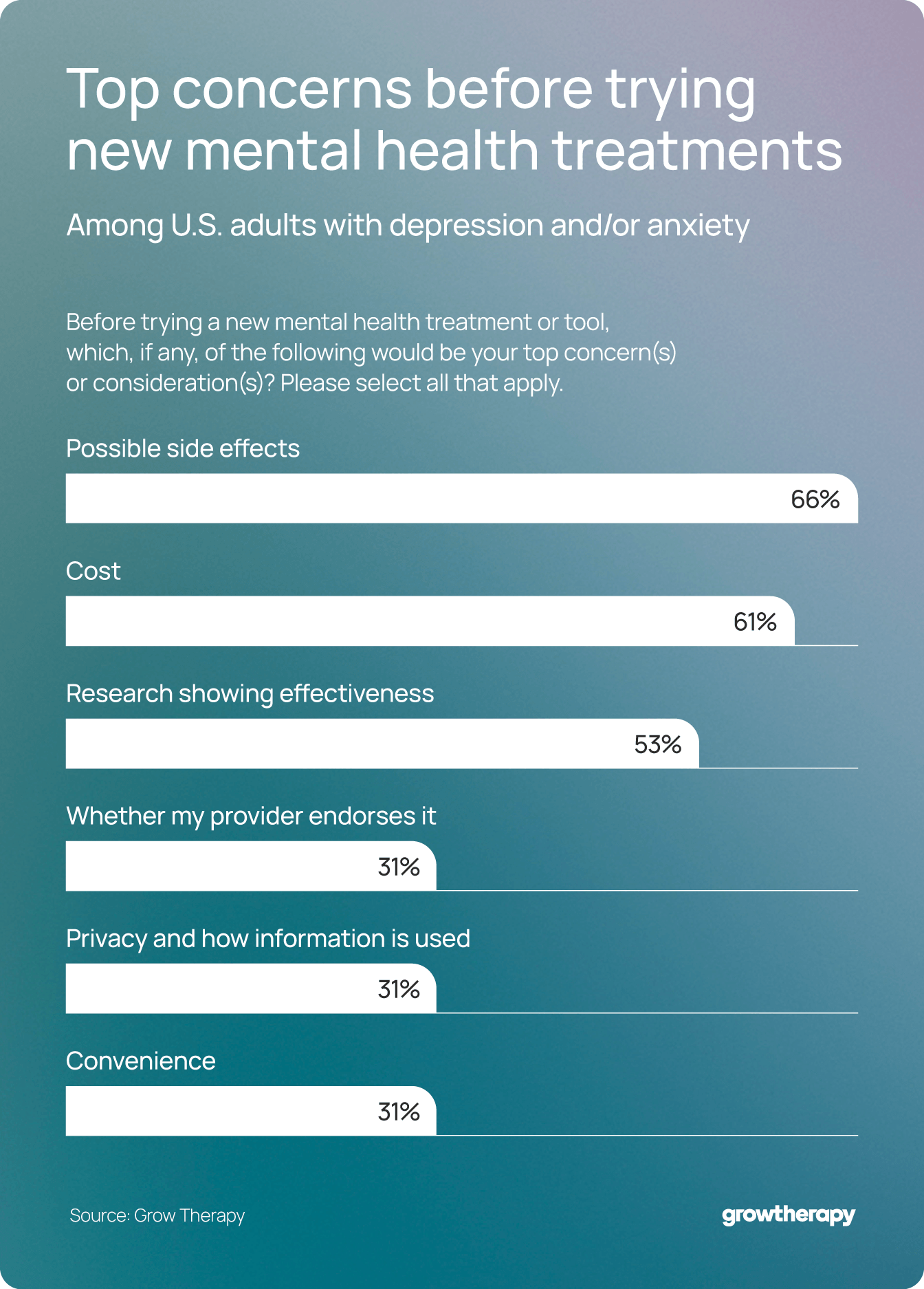
- Side effects and cost dominate decision-making: 66% of people with anxiety and/or depression say their top concern about trying new mental health treatments is possible side effects, while 61% cite cost.
- Many patients haven’t discussed newer treatment options with their providers: 56% of people with depression and/or anxiety say they have not talked with a health care provider about newly approved mental health treatments in the past 12 months.
- Confidence in sticking with digital treatment remains low: Only 15% of people with depression and/or anxiety feel very confident they could stick with a self-guided digital treatment program over time.
Types of emerging mental health treatments
Mental health treatment has expanded far beyond traditional in-person therapy. Alongside established approaches like talk therapy and medication, a growing number of newer formats are now available in clinical care — each working differently and suited to different needs. Our survey asked about these three categories in particular:
| Treatment type | Description | Examples |
|---|---|---|
| Digital support tools | App-based programs that deliver structured psychological interventions, such as cognitive behavioral therapy (CBT), via smartphone or web platforms. Often used independently or alongside traditional therapy. | DaylightRX |
| Device-based interventions | Clinical treatments that use targeted magnetic or electrical stimulation to affect brain activity in areas associated with mood. Most are administered in a provider’s office, though some devices have received clearance for supervised at-home use | NeuroStart or BrainsWay Deep TMS |
| Medication-based treatments | Medications that go beyond traditional antidepressants, sometimes offering faster-acting relief for people who haven’t responded to standard treatments. Typically administered or supervised in a clinical setting. | Spravato |
What is the difference between TMS and traditional antidepressants?
Transcranial magnetic stimulation (TMS) and antidepressant medications both treat depression, but they work in fundamentally different ways. Antidepressants are oral medications that adjust neurotransmitter levels in the brain over time — typically taking several weeks to show full effect. TMS, by contrast, is a noninvasive, device-based treatment that uses targeted magnetic pulses to stimulate specific brain regions associated with mood regulation. It is typically administered in a provider’s office over a series of sessions. TMS is generally considered for people who have not responded adequately to one or more antidepressant medications. A licensed psychiatrist or prescriber can help determine whether TMS may be appropriate based on your treatment history and clinical needs.
Possible side effects and proof matter more than cost
When considering new or unfamiliar mental health treatments — including options that may be newly available in clinical care or simply new to an individual patient — people place more weight on safety-related concerns and evidence, though cost still plays an important role.
Among adults with depression or anxiety, 66% say possible side effects are a top concern before trying a new treatment or tool. Spravato, for example, can cause dissociation, dizziness, and nausea, and must be administered in a certified clinical setting where patients are monitored for at least two hours after each dose. NeuroStar and BrainsWay Deep TMS are noninvasive but can cause temporary scalp discomfort or headaches during or after sessions, and some patients experience mild facial twitching during treatment.
A slightly smaller percentage (61%) cite cost as a top concern, while 53% cite the need for research demonstrating effectiveness.usually enjoyed, and isolation” are clear signs that a person is overloaded with responsibility and stress.
Concern about side effects also varies by age. Only 51% of Gen Z respondents cite side effects as a top consideration, compared with higher shares among older adults, suggesting comfort with newer treatment formats may differ across generations.
For providers, these concerns are an important part of the clinical conversation. Understanding what patients feel unsure about can help guide more collaborative discussions about whether a newer treatment option is clinically appropriate, relevant to their goals, and worth considering.
”Keeping track of new FDA approvals helps providers and clients understand when a mental health treatment may offer something meaningfully different, especially for people whose depression or anxiety has not improved with usual care.”
Rebecca Sturges, PMHNP-BC, psychiatry clinical quality lead at Grow Therapy
Sturges goes on to say, “Evaluating an option means looking beyond the headline and considering the person’s treatment history, likely benefits, side effects, how the treatment is given, how much monitoring it requires, and whether it fits real life.”
Accessibility is just as important as clinical fit. “Payment matters too, because insurance coverage, prior authorization, formulary rules, and reimbursement often determine whether a promising treatment is actually accessible. The goal is not just to know what is available, but to understand what is credible, appropriate, and realistically within reach for a specific person.”
Together, these findings suggest that interest in newer treatment approaches is already present, but adoption depends on whether options feel safe, are supported by evidence, and are practical in everyday care — including affordability and confidence that privacy is protected when treatment involves digital tools or remote platforms.
Many patients are open to innovation, but providers must balance patient needs and accessibility
Among adults with depression and/or anxiety, 56% say they have not discussed newly approved or advanced treatments with a provider in the past 12 months. Nearly 1 in 4 (22%) say they are not currently seeing a provider for their depression or anxiety at all. Together, these responses show many patients have not yet had an opportunity to evaluate how emerging treatment options might fit into their care.
On the other hand, 12% of people with depression and/or anxiety say a provider discussed advanced treatments and they ultimately tried at least one, while 10% say they considered treatments but chose not to pursue them.
Providers often weigh treatment history, expected benefits, monitoring requirements, and insurance coverage when deciding whether to introduce a new approach for discussion. Even when a therapy is newly approved or increasingly available, it still needs to be considered in the context of the patient’s symptoms, goals, clinical history, and current treatment plan.
Because patients often first encounter these newer options through conversations with providers, clinical guidance plays an important role in helping them understand when newer treatments may be appropriate for their needs.
Lower adoption does not necessarily reflect limited interest in newer treatment options. Instead, it highlights the role providers play in helping patients understand when emerging therapies may be appropriate and how they fit within an individual care plan. Many people first learn about newer approaches through these conversations, which helps ensure decisions are grounded in clinical evidence and real-world access considerations.
Patients report the highest openness to digital support tools among emerging treatment options
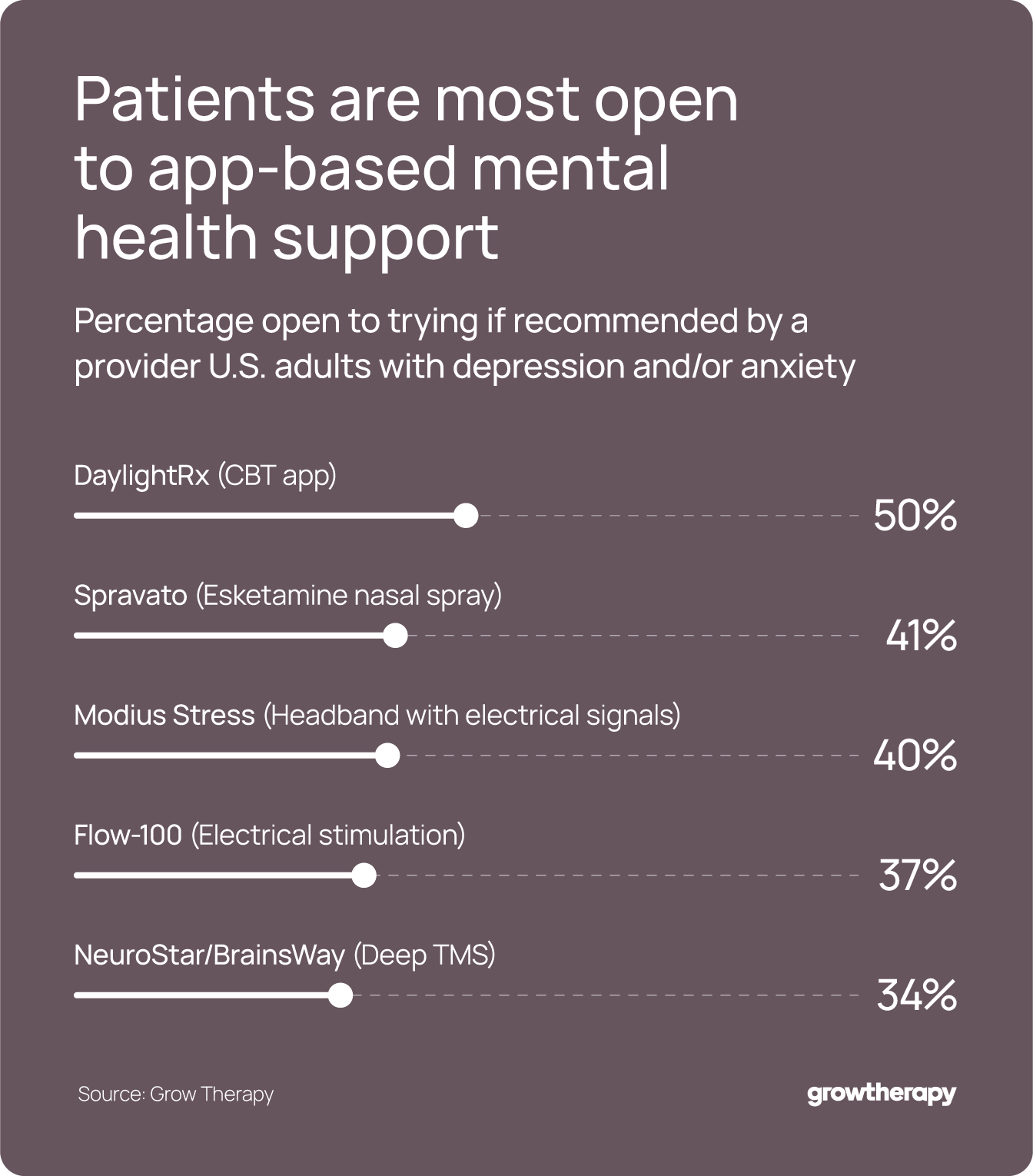
Among several emerging treatment formats included in the survey, people reported the highest openness to digital support tools compared with other newer treatment approaches.
Half (50%) of people with anxiety and/or depression are open to trying DaylightRx, a fully automated digital program that uses CBT-based techniques to help users manage worry and anxiety, which is designed to complement, not replace, traditional care.
Fewer people are open to trying Spravato (41%), an FDA-approved esketamine nasal spray for treatment-resistant depression that must be administered in a supervised clinical setting and is sometimes unfamiliar to patients because of its association with ketamine-based treatment.
Similarly, 34% say they are open to trying NeuroStar or BrainsWay Deep TMS, a noninvasive treatment that uses magnetic pulses to stimulate brain areas involved in mood regulation and is delivered through in-office device-based treatment sessions.
‘These results show that patients may view digitally delivered programs using CBT-based techniques — alongside teletherapy — as extensions of familiar care, while treatments like Spravato and deep TMS involve structured clinical visits, monitoring requirements, or specialized equipment that may impact when they are introduced into care conversations.
Confidence in digital mental health tools adherence remains a challenge
Although many patients are open to digital support tools, fewer feel confident that they could maintain engagement with self-guided programs over time.
Only 15% of people with depression and/or anxiety say they feel very confident they could maintain an at-home, self-guided digital treatment regimen for several weeks or months. More than 1 in 4 respondents (27%) express little or no confidence that they could stick with it.

The data suggest that patients may benefit from additional support when using digital mental health tools independently, particularly those managing depression or anxiety over an extended period.Approaches such as measurement-informed care strengthen that process by helping patients monitor progress and adjust treatment plans collaboratively with providers.
How patients and providers can evaluate new mental health treatment options
Mental health innovation continues to expand across digital therapy platforms and AI-assisted clinical tools. Survey responses indicate people are generally open to these advances. Introducing newer options within trusted care relationships and ensuring they are supported by clear clinical evidence are both considered best practices for supporting informed patient decision-making.
Only 12% of people with depression and/or anxiety reported that they tried advanced treatments, despite the fact that many are open to it. That gap between openness and actual use reflects a pattern seen across the findings. Low adoption doesn’t necessarily indicate little interest in or value of new therapeutic technology.
If someone’s interested in the latest mental health technology, Inder Dhillon, MD, senior medical director at Grow Therapy, advises they follow organizations like NAMI and Mental Health America, which explain major FDA approvals in plain language.
“For digital therapeutics, people should remember that not all app-based mental health treatments are validated, so it is important to look for FDA clearance, signs that a licensed clinician is involved, and research showing the app works. ClinicalTrials.gov can also be a valuable resource, especially for people seeking options beyond standard treatment,” Dhillon says.
As innovation becomes more integrated into behavioral health systems, tools that support clinicians without replacing therapeutic relationships are playing an increasingly important role. Clinical guidance and the practical realities of everyday care remain central to how patients evaluate whether newer treatment options are appropriate for them.
Talk to a provider about what’s right for you
Methodology
The survey was conducted by YouGov for Grow Therapy. The survey was fielded between February 26, 2026, and February 27, 2026. The results are based on 487 completed surveys. In order to qualify, respondents were screened to be residents of the United States, over 18 years of age, who report having depression and/or anxiety. Data is unweighted, and the margin of error is approximately +/-3% for the overall sample with a 95% confidence level.

